CASE PRESENTATION
Acute generalized exanthematous pustulosis due to paracetamol: a case report
Pustulosis exantemática generalizada aguda por paracetamol: presentación de un caso
Alvaro Rojas Mora 1*, https://orcid.org/0000-0001-6805-5364
Aylet Pérez López 1, https://orcid.org/0000-0003-4629-9995
Graciela Fundora Guerra1, https://orcid.org/0009-0000-9273-0403
1 Calixto García General University Hospital. La Habana, Cuba.
* Corresponding author: alvarorojasmorapiel@gmail.com
Received: 03/01/2026
Accepted: 22/02/2026
How to cite this article: Rojas-Mora A; Pérez-López A; Fundora-Guerra G. Acute generalized exanthematous pustulosis due to paracetamol: a case report. MedEst. [Internet]. 2026 [cited access date]; 6:e475. Available in: https://revmedest.sld.cu/index.php/medest/article/view/475
ABSTRACT
Introduction: Acute Generalized Exanthematous Pustulosis is a rare and severe cutaneous adverse reaction, primarily induced by drugs, and classified as a severe cutaneous adverse reaction. It is characterized by the sudden appearance of non-follicular, sterile pustules on an erythematous and edematous base, accompanied by fever and leukocytosis with neutrophilia.
Objective: To report a typical case of Acute Generalized Exanthematous Pustulosis due to paracetamol, confirmed using standardized diagnostic criteria.
Case presentation: We present the case of a 24-year-old male patient with Acute Generalized Exanthematous Pustulosis triggered by paracetamol. The typical clinical presentation of fever and non-follicular pustules on an erythematous base, along with compatible histopathology and a score of 11 on the European Study of Severe Cutaneous Adverse Reactions scale, confirmed the diagnosis. Management consisted of immediate discontinuation of the drug and systemic corticosteroids, with complete resolution within 3 weeks.
Conclusions: This case exemplifies that paracetamol, a commonly used medication, can induce severe skin reactions. It demonstrates the usefulness of validated scoring systems for accurate diagnosis. It reaffirms the importance of a thorough drug history in dermatology. The favorable outcome underscores that the cornerstone of treatment is the identification and withdrawal of the causative agent.
Keywords: Acute Generalized Exanthematous Pustulosis; Paracetamol.
RESUMEN
Introducción: La Pustulosis Exantemática Generalizada Aguda, es una reacción adversa cutánea grave e infrecuente y principalmente inducida por fármacos, clasificada dentro de las reacciones cutáneas adversas graves. Se caracteriza por la aparición súbita de pústulas no foliculares, estériles, sobre una base eritematosa y edematosa, acompañado de fiebre y leucocitosis con neutrofilia.
Objetivo: Reportar un caso típico de Pustulosis Exantemática Generalizada Aguda por paracetamol, confirmado mediante criterios diagnósticos estandarizados.
Presentación del caso: Presentamos el caso de un paciente masculino de 24 años con Pustulosis Exantemática Generalizada Aguda desencadenada por paracetamol. El cuadro clínico típico de fiebre y pústulas no foliculares sobre base eritematosa junto con la histopatología compatible y una puntuación de 11 puntos en la escala por el Estudio Europeo de Reacciones Cutáneas Adversas Graves, confirmaron el diagnóstico. El manejo se basó en la suspensión inmediata del fármaco y corticoides sistémicos, con resolución completa en 3 semanas.
Conclusiones: Este caso ejemplifica que el paracetamol, un medicamento de uso común, puede inducir reacciones cutáneas graves. Demuestra la utilidad de las escalas validadas para un diagnóstico preciso. Reafirma la importancia de una anamnesis farmacológica exhaustiva en dermatología. El desenlace favorable subraya que el pilar del tratamiento es la identificación y retirada del agente causal.
Palabras Clave: Pustulosis Exantemática Generalizada Aguda; Paracetamol.
INTRODUCTION
Acute Generalized Exanthematous Pustulosis (AGEP) is a rare severe adverse skin reaction (SARR), with an estimated incidence of 1–5 cases per million person-years. (1) Approximately 90 % of cases are drug-induced, with beta-lactam antibiotics, quinolones, and sulfonamides being the most frequently implicated agents. (2,3)
Clinically, it is characterized by the abrupt onset of high fever (>38°C) and a generalized eruption composed of non-follicular, sterile, subcorneal pustules on an erythematous and edematous base, typically beginning in skin folds and spreading to the trunk and extremities, with characteristic sparing of the palms and soles. (1)
Leukocytosis with neutrophilia is common, and mucosal involvement, when present, is usually mild. (2) Diagnostic confirmation is based on clinicopathological correlation, supported by the validated EuroSCAR group scale, which awards 8–12 points for definitive cases. (4)
Although paracetamol (acetaminophen) is one of the most widely used analgesics worldwide and is generally considered safe, (5) its capacity to induce severe hypersensitivity reactions, including GERD, is documented but remains under-recognized in clinical practice. (6) This condition is particularly relevant given the drug's attributed safety profile and its availability without a prescription in many countries, which can delay the identification of the causative agent and perpetuate exposure to the triggering drug.
Despite being a well-characterized condition, its low incidence leads to insufficient clinical recognition, which can result in misdiagnosis or delayed diagnosis. Therefore, the objective of this work is to report a case of Acute Generalized Exanthematous Pustulosis (AGEP) induced by paracetamol, in order to illustrate its clinical presentation, diagnostic confirmation using standardized criteria (EuroSCAR scale) and evolution after therapeutic intervention.
CASE PRESENTATION
General physical examination:
On admission, the patient was febrile (39°C), hemodynamically stable, with no signs of respiratory distress or other systemic symptoms beyond those described.
Dermatological examination:
Multiple, non-follicular, millimeter-sized pustules were observed scattered on an erythematous and edematous base on the neck, anterior and posterior chest (Figures 1 and 2). The erythema blanched with pressure. No lesions were evident on mucous membranes, palms, or soles. The remainder of the skin examination was normal.
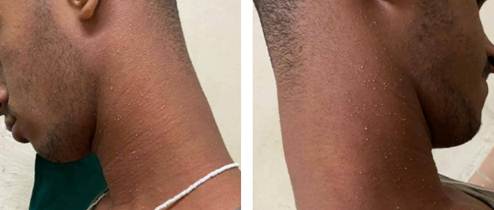
Figure 1: Anterolateral view of the neck. Multiple non-follicular, millimeter-sized pustules are observed, symmetrically distributed on an erythematous base, characteristic of the acute phase of Acute Generalized Exanthematous Pustulosis.
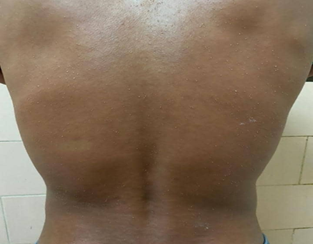
Figure 2: Posterior region of the thorax. Numerous non-follicular, millimeter-sized pustules are seen, diffusely distributed on an erythematous base.
Supplementary Examinations
Paraclinical studies revealed the following findings:
1-) Complete blood count: Leukocytosis (17.4 × 10⁹/L) with marked neutrophilia (87 %), mild eosinophilia (3 %), and relative lymphocytosis (13 %). Hematocrit: 0.44. Platelet count: 220 × 10⁹/L.
2-) Acute phase reactants: Erythrocyte sedimentation rate (ESR): 30 mm/h.
3-) Renal function: Serum creatinine: 50 µmol/L (values within normal limits).
4-) Liver function: Transaminases within normal ranges (ALT: 16 U/L, AST: 20 U/L).
5-) Serologies: HIV and general serology were non-reactive.
6-) Skin biopsy: The histopathological study showed a subcorneal pustule with abundant neutrophils, spongiosis, and neutrophilic exocytosis in the epidermis. A mixed perivascular inflammatory infiltrate, composed of neutrophils and eosinophils, was observed in the dermis. These findings were consistent with an acute hypersensitivity reaction and concordant with the proposed diagnosis.
Diagnosis: Acute Generalized Exanthematous Pustulosis (AGEP) induced by paracetamol. ICD-11 code: ED30.1. EuroSCAR score: 11 points (typical pustules: 2; typical erythema: 2; typical distribution: 2; fever >38 °C: 1; neutrophilia ≥7,000 cells/mm³: 1; compatible histology: 3), corresponding to a definitive case.
Treatment Implemented
The therapeutic management was based on the following pillars:
1- Immediate and permanent discontinuation of the causative drug: Paracetamol administration was completely discontinued.
2- Symptomatic treatment: Diphenhydramine 25 mg orally every 8 hours (for 7 days) was initiated to control pruritus.
3- Systemic corticosteroid therapy: Oral prednisone was prescribed at a dose of 1 mg/kg/day to modulate the inflammatory response.
4- Topical care: Daily emollient baths and topical application of 2% zinc oxide lotion twice daily were prescribed to protect the skin and promote healing.
Patch tests and controlled provocation tests were not performed due to the risk of recurrent reaction.
Progress and Follow-up
The patient showed a favorable clinical response. At the 12-day re-evaluation, a notable improvement was observed: complete disappearance of the pustules, marked reduction of erythema, and the presence of fine residual desquamation. A follow-up at three weeks confirmed complete resolution of the condition without evidence of cutaneous or systemic sequelae.
Informed consent: Written informed consent was obtained from the patient for the publication of this clinical case, guaranteeing the confidentiality of their identity by removing identifiable data.
DISCUSSION
Acute Generalized Exanthematous Pustulosis (AGEP) is a rare reactive dermatosis that, although classified in 1968, remains underdiagnosed in routine clinical practice. (1) The case presented illustrates the prototypical presentation of this entity: abrupt onset (<24 hours), high fever, sterile, non-follicular pustules on an erythematous base distributed on the trunk and skin folds, leukocytosis with marked neutrophilia, and a favorable outcome after discontinuation of the causative drug. (2,3)
The diagnosis of AGEP requires the exclusion of systemic pustular entities. In this case, the absence of a history of psoriasis, the atypical distribution for pustular psoriasis (sparing of the palms and soles), and the short latency period (3 days) after the start of paracetamol treatment ruled out generalized pustular psoriasis. (4) Stevens-Johnson syndrome and toxic epidermal necrolysis were excluded due to the absence of severe mucosal involvement, atypical lesions (pustules vs. macules/maculopapules), and rapid, favorable evolution. (5) Bacterial infections (staphylococcal, streptococcal) were ruled out due to the sterility of the pustules, the absence of systemic signs of infection, and the immediate response to drug withdrawal without antibiotic therapy. (6)
Confirmation using the EuroSCAR score (11/12 points) established the diagnosis with a high level of certainty. This validated tool, developed by the EuroSCAR group, has demonstrated 98% specificity and 80 % sensitivity for discriminating PEGA from other SGRTs. (6) The score obtained far exceeds the "definite case" threshold (≥8 points), reinforcing the validity of the report.
Although beta-lactam antibiotics account for 40–50 % of pharmacodynamic paracetamol-induced hypersensitivity reactions (PINs), paracetamol is identified as the cause in approximately 3–5 % of cases according to European registries. The relevance of this association lies in the safety profile attributed to the drug and its over-the-counter status in multiple countries, including Cuba. (8–10)
The observed latency (3 days from the start of paracetamol administration to the onset of symptoms) is characteristic of immediate T-cell-mediated hypersensitivity reactions (type IVc reaction according to the modified Gell and Coombs classification). This immunological mechanism, distinct from direct toxicity by hepatotoxic metabolites (N-acetyl-p-benzoquinone imine), explains the individuality of the response and the impossibility of predicting it using therapeutic doses. (11) The presentation at first documented exposure suggests prior subclinical sensitization or a heteroallergic reaction due to shared antigenic determinants. (12,13,14)
The choice of oral systemic corticosteroid therapy (prednisone 1 mg/kg/day) was based on the extent of the skin lesion (>50% body surface area) and significant systemic discomfort. (15,16) Although AGE is self-limiting, corticosteroids accelerate resolution by approximately 3–5 days according to observational evidence. (5) Intravenous corticosteroid therapy and prolonged hospitalization were not indicated given the patient's hemodynamic stability, absence of organ involvement, and adequate family compliance.
The decision not to perform patch testing or controlled oral provocation testing was justified by: (a) a definitive clinicopathological diagnosis, (b) the risk of potentially severe recurrence, and (c) the availability of effective analgesic alternatives. (17) This decision is consistent with recommendations from European dermatological societies for cases with a high degree of diagnostic certainty. (6)
This case underscores the need for reporting systems for adverse drug reactions to over-the-counter medications. Paracetamol, consumed by 80 % of the Cuban adult population at least once a year, generates a false sense of security that delays the diagnosis of serious reactions. A thorough medication history should explicitly include "self-medication" and products considered "harmless." (18)
Reporting this case to the National Pharmacovigilance Center contributes to characterizing the actual safety profile of paracetamol and strengthens post-marketing surveillance of widely used medications.
CONCLUSIONS
This case confirms the diagnosis of Acute Generalized Exanthematous Pustulosis (AGEP) induced by paracetamol, validated by the EuroSCAR score, and illustrates the capacity of commonly used drugs to trigger severe hypersensitivity reactions. The favorable outcome after discontinuation of the causative agent and timely systemic treatment reaffirms the self-limiting nature of the condition. This report reinforces the need for a thorough medication history and contributes to pharmacovigilance, reminding us that no drug is free from serious adverse risks.
BIBLIOGRAPHIC REFERENCES
1. Saavedra S, Briceño M, Pajuelo G, Sandoval B. Pustulosis exantematosa generalizada aguda. An Fac Med (Lima). 2021;82(4):322-5. DOI:10.15381/anales.v82i4.21025
2. Puebla-Miranda M, González-Rivera C, Reséndiz-Carmona G, Vázquez-Velo JA. Pustulosis exantemática aguda generalizada. Dermatol Rev Mex. [Internet] 2023 [cited 02/01/2026]; 67(4):536-42. Available in: https://dermatologiarevistamexicana.org.mx/article/pustulosis-exantematica-aguda-generalizada/
3. Moore MJ, Sathe NC, Ganipisetti VM. Pustulosis exantemática generalizada aguda [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 [cited 02/01/2026]. Available in: https://www.ncbi.nlm.nih.gov/books/NBK592407/
4. Sapia E, Lascano F, García Zubillaga P, Dastugue M. Pustulosis exantemática aguda generalizada. Rev Hosp Niños (B Aires). [Internet]. 2019 [cited 02/01/2026]; 61(274):159-64. Available in: https://revfdc.sld.cu/index.php/fdc/article/view/190
5. Lizardo-Castro GA, Guillén-Mejía GW. Acute generalized exanthematous pustulosis. Bol Med Hosp Infant Mex. 2022;79(4):268-73. DOI:10.24875/BMHIM.22000268
6. Stadler P, Oschmann A, Kerl K, Maul J, Oppel E, Schiesser B, et al. Acute generalized exanthematous pustulosis: clinical characteristics, pathogenesis, and management. Dermatology. 2023;239:328-33. DOI:10.1159/000529218
7. Alfalah M, Alotaibi Y, Alotaibi A, Alharthi R. Acute generalized exanthematous pustulosis induced by iodinated contrast media: a case report. Dermatol Reports. 2025;17(4):10217. doi:10.4081/dr.2025.10217
8. Tejera Díaz JF, González González L, Hurtado Gascón L, Portela Arrieta MC, Delgado Silva M, Cansino Carvajal L, et al. Pustulosis exantemática generalizada aguda. Folia Dermatol Cubana [Internet]. 2023 [cited 02/01/2026]; 16(1). Available in: https://revfdc.sld.cu/index.php/fdc/article/view/307
9. Vallejo-Yagüe E, Martinez-De la Torre A, Mohamad OS, Sabu S, Burden AM. Desencadenantes farmacológicos y cuadro clínico de la pustulosis exantemática aguda generalizada (PEAG): una serie de casos de 297 pacientes. J Clin Med. 2022;11(2):397. DOI:10.3390/jcm11020397
10. Creadore A, Desai S, Alloo A, Dewan AK, Bakhtiar M, Cruz-Diaz C, et al. Clinical Characteristics, Disease Course, and Outcomes of Patients With Acute Generalized Exanthematous Pustulosis in the US. JAMA Dermatol. 2022;158(2):176-183. doi:10.1001/jamadermatol.2021.5390
11. Hadavand MA, Kaffenberger B, Cartron AM, Trinidad JCL. Clinical presentation and management of atypical and recalcitrant acute generalized exanthematous pustulosis. J Am Acad Dermatol. 2022;87(3):632-639. doi:10.1016/j.jaad.2020.09.024
12. de Groot AC. Results of patch testing in acute generalized exanthematous pustulosis (AGEP): A literature review. Contact Dermatitis. 2022;87(2):119-141. doi:10.1111/cod.14075
13. Castanedo-Lara LM, Sánchez-Olivo N. Pustulosis exantemática aguda generalizada inducida por metotrexato. Reacción adversa infrecuente pero potencialmente grave. Dermatol Rev Mex. [Internet]. 2025 [cited 02/01/2026]; 69(6):930-2. Available in: https://dermatologiarevistamexicana.org.mx/article/pustulosis-exantematica-aguda-generalizada-inducida-por-metotrexato-reaccion-adversa-infrecuente-pero-potencialmente-grave/
14. Gómez-Chicre V, Camacaro MG, Guijarro J, López de la Torre M, Martínez Díaz N, Abadía L. Pustulosis exantemática generalizada aguda en paciente pediátrico polimedicado con traumatismo craneoencefálico. Rev Asoc Colomb Dermatol. 2022;30(1):52-9. DOI:10.29176/2590843X.1730
15. Parisi R, Shah H, Navarini AA, Serra-Baldrich E, Veraldi S, Rieder F, et al. Acute generalized exanthematous pustulosis: clinical features, differential diagnosis, and management. Am J Clin Dermatol. 2023;24(4):557-75. DOI:10.1007/s40257-023-00779-3
16. Santos MI, Sousa M, Cerqueira P, Ambrioso I, Moniz Ramos M. A case of acute generalized exanthematous pustulosis induced by ceftriaxone. Cureus. 2023;15(3):e36281. DOI:10.7759/cureus.36281
17. Shi D, Ghias M, Haris A, Heubach K. Acute generalized exanthematous pustulosis in a patient with Crohn's disease being treated for streptococcal pharyngitis and sternoclavicular joint abscess. Cureus. 2025;17(1):e77595. DOI:10.7759/cureus.77595
18. Vo VA, Dupuy E, Rukasin CRF. Combination testing confirming clindamycin induced acute generalized exanthematous pustulosis. JAAD Case Rep. 2025;63:77-80. DOI:10.1016/j.jdcr.2025.06.005
AUTHORSHIP CONTRIBUTION
ARM: Conceptualization, research, data curation, methodology, visualization, drafting of the original manuscript, and revision and editing of the final manuscript.
ALP: Conceptualization, research, and supervision.
GFG: Conceptualization, research, and supervision.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
FUNDING SOURCES
The authors received no funding for the development of this article.
USE OF ARTIFICIAL INTELLIGENCE
The authors declare that no artificial intelligence was used in the writing of this manuscript.