CASE PRESENTATION
Multifocal mixed testicular cancer with osteoblastic bone metastases: a case report
Cáncer testicular multifocal mixto con metástasis óseas osteoblásticas: presentación de un caso
Keily Alvarez Gómez 1, https://orcid.org/0000-0003-1709-1837
Jean Marcos Campanería Hernández 1, https://orcid.org/0009-0008-2065-6855
Carlos Luis Vinageras Hidalgo 1, https://orcid.org/0009-0007-4848-1797
José Antonio Méndez Fiallo 2, https://orcid.org/0000-0003-0616-9862
1 University of Medical Sciences of Matanzas. Faculty of Medical Sciences of Matanzas “Dr. Juan Guiteras Gener”, Matanzas. Cuba.
2 “Faustino Pérez” Provincial Clinical-Surgical-Teaching Hospital. Matanzas, Cuba.
* Corresponding author: kikialva04@gmail.com
Received: 03/03/2026
Accepted: 08/04/2026
Published: 23/04/2026
How to cite this article: Alvarez-Gómez K, Campanería-Hernández JM, Vinageras-Hidalgo CL, Méndez-Fiallo JA. Multifocal mixed testicular cancer with osteoblastic bone metastases: a case report. MedEst. [Internet]. 2026 [citado acceso fecha]; 6:e533. Disponible en: https://revmedest.sld.cu/index.php/medest/article/view/533
ABSTRACT
Introduction: Testicular cancer is the most common neoplasm in men aged 15 to 35. Mixed germ cell tumors account for up to 60% of non-seminomatous cases. Bone dissemination is exceptional (3–9%), and osteoblastic metastases are extremely rare, associated almost exclusively with predominant choriocarcinoma.
Objective: To describe a rare case of multifocal mixed testicular cancer with osteoblastic bone metastases.
Case Presentation: A 46-year-old male patient presented with a 1 cm, hard, painless mass in the left testicle, present for 3 months. Tumor markers showed normal alpha-fetoprotein levels (1.53 IU/mL) and elevated β-hCG levels (13.4 IU/mL). Doppler ultrasound revealed a heterogeneous, hypervascularized mass measuring 10 × 8 mm. Computed tomography with bone window settings revealed osteoblastic lesions in the thoracic spine and the left femoral head. A left inguinal orchiectomy was performed. Histological analysis reported a mixed germ cell tumor consisting of seminoma (50%), embryonal carcinoma (25%), yolk sac tumor (15%), and choriocarcinoma (10%), with lymphovascular invasion.
Conclusions: Mixed testicular cancer containing choriocarcinoma (10%) may present with osteoblastic bone metastases—an exceptional finding that justifies the use of bone window settings during staging. Normal alpha-fetoprotein levels do not rule out metastatic disease when β-hCG levels are elevated. Reporting such atypical presentations enhances clinical suspicion in patients falling outside the classic age range for this disease.
Keywords: Testicular Cancer; Germ Cells; Bone Metastasis; Multimodal Treatment.
RESUMEN
Introducción: el cáncer testicular es la neoplasia más frecuente en hombres de 15 a 35 años. Los tumores de células germinales mixtos representan hasta el 60% de los casos no seminomatosos. La diseminación ósea es excepcional (3-9%) y las metástasis osteoblásticas son extremadamente infrecuentes, asociadas casi exclusivamente a coriocarcinoma predominante.
Objetivo: describir un caso infrecuente de cáncer testicular multifocal mixto con metástasis óseas osteoblásticas.
Presentación del caso: paciente masculino de 46 años, con masa testicular izquierda de 1 cm, dura e indolora de 3 meses de evolución. Los marcadores tumorales mostraron alfafetoproteína normal (1,53 UI/ml) y β-hCG elevada (13,4 UI/ml). La ecografía Doppler evidenció una masa heterogénea hipervascularizada de 10×8 mm. La tomografía computarizada con ventana ósea reveló lesiones osteoblásticas en columna dorsal y cabeza femoral izquierda. Se realizó orquiectomía inguinal izquierda. La histología informó tumor de células germinales mixto: seminoma 50%, carcinoma embrionario 25%, tumor del saco vitelino 15% y coriocarcinoma 10%, con invasión linfovascular.
Conclusiones: el cáncer testicular mixto con coriocarcinoma (10%) puede presentar metástasis óseas osteoblásticas, un hallazgo excepcional que justifica ventana ósea en la estadificación. La normalidad de la alfafetoproteína no excluye enfermedad metastásica cuando la β-hCG está elevada. Reportar estas presentaciones atípicas mejora la sospecha clínica fuera del rango etario clásico.
Palabras clave: Cáncer De Testículo; Células Germinales; Metástasis Ósea; Tratamiento Multimodal.
INTRODUCTION
Testicular cancer represents less than 1% of all male cancers; however, it is the most common malignancy in the age group from 15 to 35 years. Currently, the five-year survival rate exceeds 95%, thanks to the effectiveness of combining surgery and platinum-based chemotherapy (1). About 90-95% of these cases are germ cell tumors (GCT), and among them, mixed histological types represent between 32% and 60% of non-seminoma tumors (2).
Multifocality — defined as having two or more tumor areas separated by more than 1 mm within the same testicle — is reported in a variable proportion (15.7% to 33%) and is often linked to lesions smaller than 2 cm (2). Although GCT typically spreads through the lymphatic system to the retroperitoneum, blood-borne metastases can affect the lungs, liver, and brain (3). In contrast, bone involvement is rare, with reported incidence of 3% at diagnosis and up to 9% at the time of recurrence (4); these lesions are mostly lytic (5).
Osteoblastic presentation is extremely unusual and has been mainly linked to pure choriocarcinoma or tumors where this component is dominant (6). This case stands out for its clinical uniqueness due to the combination of three atypical factors: a 46-year-old patient (outside the usual age range), a multifocal mixed germ cell tumor, and the presence of osteoblastic bone metastases. Also, there was a marked difference between serum tumor markers (normal AFP and high β-hCG) and the final complex histology (seminoma 50%, embryonal carcinoma 25%, yolk sac tumor 15% and choriocarcinoma 10%), which creates a diagnostic and therapeutic challenge of high educational value (6, 7).
For this reason, the aim of this paper is to describe a rare case of multifocal mixed testicular cancer with osteoblastic bone metastases.
CASE PRESENTATION
A 46-year-old male patient with a history of heavy smoking and regular alcohol use. No personal or family history of cancer. He came to the Urology clinic reporting that about three months earlier he had noticed a mass in his left testicle, about 1 cm in diameter, which he described as hard, painless, attached to deeper tissues, and not significantly growing in size. He denied pain, fever, previous injury, changes in urination, erectile dysfunction, weight loss, night sweats, or any general symptoms.
On physical examination, the patient had no fever, was hemodynamically stable, and had vital signs within normal limits. Genital examination showed a normal penis and retractable foreskin. The right testicle was normal in size, shape, and consistency. The left testicle had a palpable tumor mass of approximately 1 cm, hard, non-tender, attached to deeper tissues, with no local signs of inflammation. The abdomen was soft, not painful to superficial or deep touch, with no palpable masses or groin or abdominal lymph nodes.
Additional tests
Initial lab results showed: hemoglobin 13.6 g/dL, hematocrit 42%, white blood cells 10.0 × 10⁹/L, platelets 280 × 10⁹/L, ESR 10 mm/h, glucose 4.4 mmol/L, creatinine 84.2 µmol/L, total cholesterol 4.5 mmol/L, triglycerides 1.0 mmol/L, alkaline phosphatase 405.3 U/L (high), and AST 57.3 U/L.
Serum tumor markers: alpha-fetoprotein (AFP) 1.53 IU/ml (normal range), beta-human chorionic gonadotropin (β-hCG) 13.4 IU/ml (high).
Scrotal Doppler ultrasound (Figure 1): showed a mixed, hypoechoic tumor mass located in the front part and upper pole of the left testicle, measuring 10 × 8 mm, with cystic areas inside and tiny calcifications. Color Doppler study showed increased internal blood flow (highly vascular), a pattern suggestive of germ cell tumor (Figure 2). On cross-sectional imaging, the solid nature of the lesion was seen, with irregular edges and no posterior acoustic shadow (Figure 3).

Figure 1: Scrotal Doppler ultrasound, longitudinal view. Shows a mixed hypoechoic mass at the upper pole of the left testicle (arrow), with internal cystic areas and tiny calcifications. Maximum diameter: 10 mm.
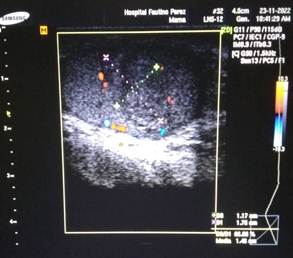
Figure 2: Color Doppler ultrasound, same patient. The mass shows increased internal blood flow (highly vascular), a pattern suggestive of germ cell tumor.
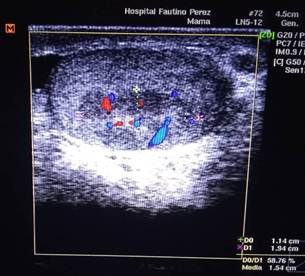
Figure 3: Transverse ultrasound view. Shows the solid nature of the lesion, with irregular edges and no posterior acoustic shadow.
Abdominal ultrasound: No abnormalities of intra-abdominal organs or visible lymph nodes.
Chest X-ray: No bone or lung abnormalities.
CT scan of chest, abdomen, and pelvis with bone windows: No pleuropulmonary abnormalities were seen. Bone windows showed osteoblastic lesions in the thoracic spine and left femoral head, suggestive of bone metastases.
ICD-11 classification: Multifocal mixed germ cell testicular cancer: 2C80.0 (Malignant tumor of testis). Bone metastasis: 2D8Z (Metastasis of malignant neoplasm of unspecified site)
Surgical procedure and intraoperative findings
A left inguinal orchiectomy with high ligation of the spermatic cord was performed (Figure 4). During surgery, the left testicle was seen to be enlarged and had lost its normal consistency. When the surgical specimen was opened (Figure 5), the multifocal tumor was seen replacing the testicular tissue, with areas of necrosis and bleeding consistent with choriocarcinoma.
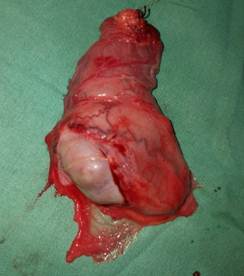
Figure 4: Intraoperative finding during left inguinal orchiectomy. The spermatic cord is exposed before high ligation. Left testicle appears enlarged and has lost its normal consistency.
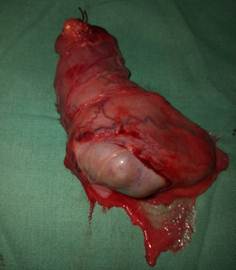
Figure 5: Opened orchiectomy specimen. The multifocal tumor can be seen replacing testicular tissue, with areas of necrosis and bleeding consistent with choriocarcinoma.
Final histological report
· Multifocal tumor with the largest nodule measuring 1.8 × 2.0 cm.
· Mixed germ cell tumor with the following composition: Classic seminoma: 50%, Embryonal carcinoma: 25%, Yolk sac tumor: 15%, Choriocarcinoma: 10%.
· Tumor spread: infiltration of the rete testis and tunica albuginea.
· Lymphovascular invasion present.
· Other findings: presence of germ cells in situ and Leydig cell hyperplasia.
Treatment and follow-up
After surgery, the patient received systemic chemotherapy with a regimen not specified in the medical record (reported as one monthly session for four months). The prognosis at discharge was guarded. At the time of writing this manuscript, the patient has finished cancer treatment and is waiting for follow-up with new tumor markers and a repeat CT scan.
Informed consent
Written informed consent was obtained from the patient for the publication of this case report, ensuring the confidentiality of his identity by removing any identifiable data. The patient also explicitly authorized the use of surgical and ultrasound images for scientific and educational purposes.
DISCUSSION
This case involves a 46-year-old patient diagnosed with multifocal mixed testicular cancer (seminoma 50%, embryonal carcinoma 25%, yolk sac tumor 15% and choriocarcinoma 10%) who first presented with osteoblastic bone metastases in the thoracic spine and left femoral head, despite having a small primary testicular mass (10 × 8 mm). This finding is exceptional for several reasons, which are discussed below.
Bone metastases from testicular germ cell tumors are uncommon. The NCCN (2024) guidelines report an incidence of 3% at initial presentation and 9% at recurrence (4). However, the vast majority of these metastases are lytic (5). Osteoblastic presentation is extremely rare and has been described almost exclusively in pure choriocarcinoma or in tumors where this component is dominant (6). In a review of the literature, Sia and colleagues (2022) identified exceptional cases of osteoblastic bone metastases in mixed germ cell tumors, highlighting that even small amounts of choriocarcinoma can lead to aggressive biological behavior (6). The case presented here differs from other reports because the choriocarcinoma component represents only 10% of the tumor composition, suggesting that even minor proportions of this subtype can determine an atypical pattern of spread (6, 7, 8).
Regarding age, the patient was 46 years old, above the usual peak incidence (15-35 years) (1). Although testicular cancer can occur in older adults, the literature indicates that mixed germ cell tumors in patients over 40 have different clinicopathological features, with a higher likelihood of atypical spread (9). Recent epidemiological studies project an increase in the incidence of this cancer in older age groups, which means we must maintain a high level of suspicion beyond the classic range (5).
One particularly relevant finding is the mismatch between serum tumor markers and the histological makeup. Alpha-fetoprotein (AFP) was within normal range (1.53 IU/ml), even though the yolk sac tumor made up 15% of the cancer. β-hCG was elevated (13.4 IU/ml), which is expected due to the choriocarcinoma component (10%). However, normal AFP in the presence of a yolk sac tumor has been reported in up to 30-40% of cases, especially when the component is minor (<20%) or when there are issues with the production or secretion of the glycoprotein (6). The current World Health Organization (2022) classification emphasizes that the correlation between serum markers and histology is not perfect, and that immunohistochemistry of the tissue may reveal significant mismatches (7, 8). This phenomenon can lead to underestimating tumor aggressiveness and delay the decision to start systemic treatment (6).
In this case, the diagnosis of bone metastases was made using a CT scan with bone windows, a study that is not part of the usual staging protocol (4). This raises the question of whether other patients with a similar mismatch could benefit from a broader diagnostic approach, especially when alkaline phosphatase is high (405.3 U/L in this patient), a finding that should prompt a targeted search for bone metastases (6).
The surgical approach was appropriate: inguinal orchiectomy with high ligation of the spermatic cord is the gold standard for the diagnosis and local treatment of testicular cancer (2). The 2023 European Association of Urology (EAU) guidelines recommend this procedure as the primary treatment for all testicular germ cell tumors (2). However, potential areas for improvement in management were identified:
- Initial staging: Although a CT scan of the chest, abdomen, and pelvis was performed, it is not specified whether intravenous contrast was used to properly evaluate the retroperitoneum. The presence of bone metastases without detectable retroperitoneal disease is unusual and should have prompted a targeted search for blood-borne spread (6).
- Chemotherapy regimen: The medical record only states "one monthly session for four months," without specifying the regimen used. For a non-seminoma germ cell tumor with visceral (bone) metastases, the risk classification according to the International Germ Cell Cancer Collaborative Group (IGCCCG) is poor risk, and the standard treatment is four cycles of BEP chemotherapy (bleomycin, etoposide, cisplatin) (4). The absence of this information limits the reproducibility of the case and the evaluation of treatment response.
- Follow-up: At the time of this report, the patient was waiting for new tumor markers and a repeat CT scan. There is no information on whether he completed the four cycles, whether he experienced toxicity, or what the radiological or biochemical response was. Current protocols recommend close follow-up with tumor markers every 2-3 months and imaging every 6-12 months during the first two years (4).
The presence of osteoblastic bone metastases from a testicular germ cell tumor should not automatically rule out a primary testicular cancer, even in patients over 40 with a small testicular mass (6).
The mismatch between tumor markers and histology (normal AFP with yolk sac tumor present) does not rule out aggressiveness, and should prompt more thorough staging, including bone windows in imaging studies (6, 7, 8).
The choriocarcinoma component, even in a low proportion (10%), can lead to an atypical metastatic potential, especially to bone. The 2022 WHO classification recognizes that the histological makeup, beyond the dominant subtype, determines biological behavior (7, 8).
It is essential to clearly record the chemotherapy regimen used, as well as the IGCCCG risk classification, to allow comparison with other cases and guide future treatment (4).
This report has limitations inherent to its descriptive nature: the exact chemotherapy regimen is not available, complete post-treatment follow-up is not available, and no molecular studies (immunohistochemistry for β-hCG and AFP on tissue) were performed that could have explained the marker mismatch (7, 8). However, despite these limitations, the case provides educational value by documenting an atypical presentation that should be considered in daily clinical practice.
CONCLUSIONS
Multifocal mixed testicular cancer with choriocarcinoma (10%) can present with osteoblastic bone metastases — an exceptional finding that justifies the use of bone windows on staging CT scans. Normal alpha-fetoprotein levels, despite the presence of yolk sac tumor (15%), do not rule out metastatic disease when β-hCG is elevated. Inguinal orchiectomy is the standard surgical treatment, but in patients with bone metastases and over 40 years old, the poor risk classification requires four cycles of BEP chemotherapy. Reporting these atypical presentations improves clinical suspicion outside the classic age range.
BIBLIOGRAPHIC REFERENCES
1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71(3):209-49. https://doi.org/10.3322/caac.21660
2. Laguna MP, Albers P, Algaba F, Bokemeyer C, Boormans JL, Fischer S, et al. EAU Guidelines on Testicular Cancer [Internet]. Arnhem: European Association of Urology; [Internet] 2023 [cited 03/03/2026]. Available at: https://uroweb.org/guidelines/testicular-cancer
3. Goswami PR, Singh G, Patel T, Dave R. The WHO 2022 Classification of Renal Neoplasms (5th Edition): Salient Updates. Cureus. 2024;16(4):e58470. doi: 10.7759/cureus.58470.
4. National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Testicular Cancer. Version 2.2022 [Internet]. Plymouth Meeting: NCCN; 2022 [cited 03/03/2026]. Available at: https://www.nccn.org/guidelines
5. Ghazarian AA, Kelly SP, Altekruse SF, Rosenberg PS, McGlynn KA. Future of testicular germ cell tumor incidence in the United States: Forecast through 2026. Cancer. 2017 Jun 15;123(12):2320-2328. doi: 10.1002/cncr.30597.
6. Oldenburg J, Berney DM, Bokemeyer C, Climent MA, Daugaard G, Gietema JA, et al. Testicular seminoma and non-seminoma: ESMO-EURACAN Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2022 Apr;33(4):362-375. doi: 10.1016/j.annonc.2022.01.002.
7. Znaor A, Skakkebaek NE, Rajpert-De Meyts E, Laversanne M, Kuliš T, Gurney J, Sarfati D, McGlynn KA, Bray F. Testicular cancer incidence predictions in Europe 2010-2035: A rising burden despite population ageing. Int J Cancer. 2020 Aug 1;147(3):820-828. doi: 10.1002/ijc.32810.
8. Rajpert-De Meyts E, Aksglaede L, Bandak M, Toppari J, Jørgensen N. Testicular Cancer: Pathogenesis, Diagnosis and Management with Focus on Endocrine Aspects. In: Feingold KR, Adler RA, Ahmed SF, Anawalt BD, Blackman MR, Boyce A, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000-. [Last update: 2023 Mar 29; cited 2026 mar 03]. Available at: https://www.ncbi.nlm.nih.gov/books/NBK278992/
9. Stang A, Jöckel KH, Baumgardt-Elms C, Ahrens W, Bromen K, Johnen G, et al. Incidence of testicular tumor subtypes according to the updated WHO classification. Andrology. 2019;7(4):402-407. doi: 10.1111/andr.12565.
10. Dubey H, Jain G, Kumar C, Ranjan A, Batra A, Chellapuram SK, Gupta S, Goel H, Sharma A, Tanwar P. Bone marrow metastasis of testicular germ cell tumour: A rare case. Heliyon. 2023 May 29;9(6):e16703. doi: 10.1016/j.heliyon.2023.e16703.
11. Tateo V, Thompson ZJ, Gilbert SM, Cortessis VK, Daneshmand S, Masterson TA, Feldman DR, Pierorazio PM, Prakash G, Heidenreich A, Albers P, Necchi A, Spiess PE. Epidemiology and Risk Factors for Testicular Cancer: A Systematic Review. Eur Urol. 2025 Apr;87(4):427-441. doi: 10.1016/j.eururo.2024.10.023.
AUTHOR CONTRIBUTIONS
KAG: Conceptualization, investigation, data curation, methodology, visualization, writing of the original draft, as well as review and editing of the final manuscript.
JMCH: Conceptualization, investigation, and supervision.
CLVH: Conceptualization, investigation, data curation, and editing of the final manuscript.
JAMF: Conceptualization, investigation, supervision, and editing of the final manuscript.
CONFLICT OF INTERESTS
The authors declare no conflict of interest.
FUNDING SOURCES
The authors received no funding for the development of this article.
USE OF ARTIFICIAL INTELLIGENCE
The authors declare that no artificial intelligence was used in the writing of this manuscript.